|
As all of the other substances are soluble in water we can rewrite the equation. The most common examples of precipitation are Rain, Snowfall, hail, sleet, dew etc. A double displacement precipitation is a reaction in which two soluble ionic compounds react to form an insoluble precipitate. Because it is insoluble in water we know that it is the precipitate. Answer: The precipitation occurs in nature. In a double-replacement reaction, the cations (positively charged species) or anions (negatively charged species) of each compound swap places, forming new bonds/products. We would expect them to undergo a double displacement reaction with each other.īy examining the solubility rules we see that, while most sulfates are soluble, barium sulfate is not. Precipitate usually means 'bringing something on' or 'making it happen' and not always in a good way. A precipitation reaction is a special kind of double-replacement reaction where two soluble compounds react to form an insoluble solid called a precipitate. 
Write the reaction and identify the precipitate.īarium chloride and potassium sulfate are both ionic compounds. The chemical reaction occurs as follows: Ag + (aq) + Cl (aq) AgCl (s). Example To determine the concentration of chloride ion in a certain solution we can titrate this solution with silver nitrate solution (whose concentration is known). 
The exceptions are the alkali metals and the ammonium ion.ĬaSO 4 and Ag 2SO 4 are slightly soluble.Ī solution of barium chloride is mixed with a solution of potassium sulfate and a precipitate forms. Here we have discussed an example of precipitation titration. Carbonates (CO 3 -2), phosphates (PO 4 -3) and sulfides (S -2) are insoluble. The exceptions are those containing Ag +, Hg +2, and Pb +2.Ħ. Most chlorides (Cl -), bromides (Br -) or iodides (I -) are soluble. The exceptions are the alkali metal hydroxides and Ba(OH) 2.ĥ. of 6.37 for the first deprotonation of carbonic acid ( ), what is the ratio of bicarbonate () to carbonic acid (. How many grams of precipitate form and this reaction produces 18.7 g precipitate. Students then carry out a bubble test on the solid soap scum and Ivory soap to determine if they are different. A chemist combines 300 mL of a 0.3 M solution with 200 mL of 0.4 M solution. Nitrates (NO 3 -), chlorates (ClO 3 -), and perchlorates (ClO 4 -) are soluble. Students combine the soap solution with the Epsom salt solution to produce a solid soap scum precipitate. Ammonium (NH 4 +) compounds are soluble.ģ. 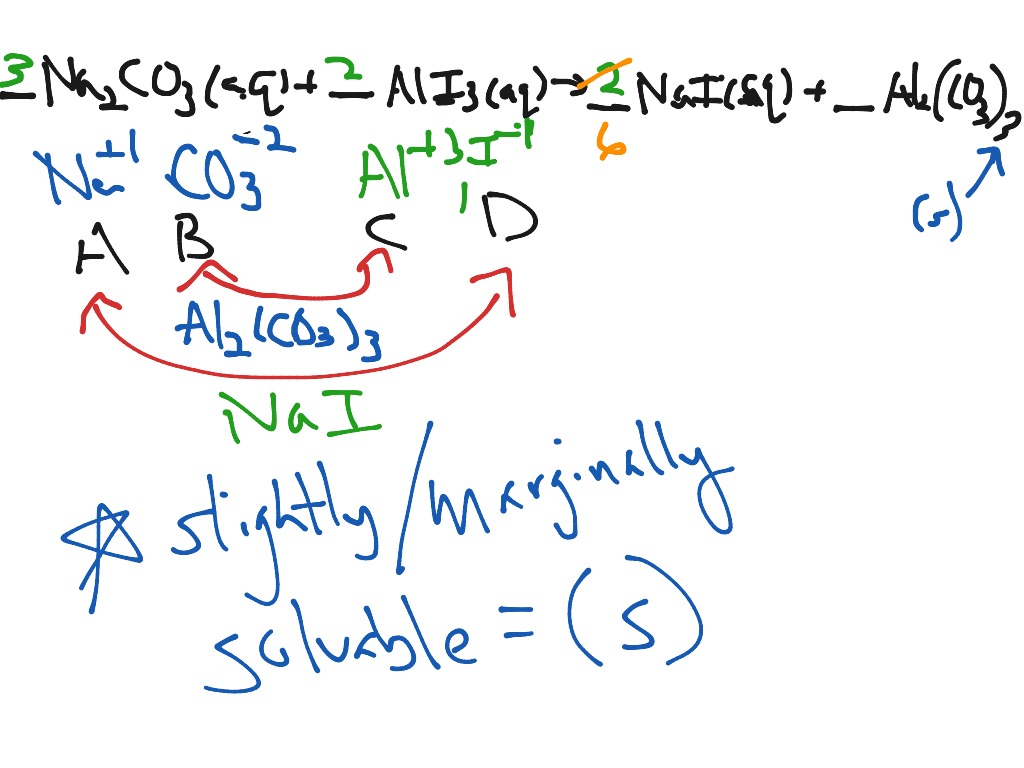
Alkali metal (Group IA) compounds are soluble.Ģ. Solubility Rules and Identifying a Precipitateġ. Examples of precipitate deposits that can cause clogged pipes include magnesium and calcium oxides that result from hard water formation.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
